
DengYue Medicine
Member Info
-
Profile Type: Regular Member
-
Profile Views: 491 views
-
Friends: 0 friends
-
Last Update: Mar 31
-
Last Login: Mar 31
-
Joined: November 21, 2025
-
Member Level: Default Level
Updates

Nothing has been posted here yet - be the first!
Info
Personal Information
- First Name DengYue
- Last Name Medicine
Contact Information
Personal Details
- About Me DengYueMed is a Hong Kong-based pharmaceutical wholesaler specializing in the export of Chinese medicines. With a product portfolio of over 30,000 types, our offerings include Chinese innovative drugs, oncology and hematology treatments, orphan medicines, and a wide range of hospital-use pharmaceuticals. Through an extensive global trade network, we are committed to ensuring the worldwide supply of high-quality medicines to meet diverse healthcare needs. We consistently prioritize the safety, efficacy, and stable quality of our products, ensuring that all medicines comply with international certification standards.
Blogs
Forum Posts
-
- DengYue Medicine
- 2 posts
Posted in the topic DengYueMed: Independent Global Pharmaceutical Intelligence Platform – Empowering Insights into Oncology, Rare Diseases, and Regulatory Frontiers in the forum Off-Topic DiscussionsMarch 31, 2026 7:30 PM PDTIn the fast-evolving global pharmaceutical landscape of 2026, companies, investors, and clinicians need timely, unbiased, and actionable intelligence more than ever. DengYueMed stands out as a dedicated independent platform focused on global pharmaceutical intelligence, with deep expertise in Oncology, Rare Diseases, Regulatory Affairs, and Supply Chain dynamics. Covering the FDA, EMA, and NMPA across the US, Europe, and China, it delivers in-depth analysis and practical insights that bridge innovation, market access, cross-border distribution, and policy shifts.
Unlike traditional consulting reports or corporate-sponsored platforms, DengYueMed offers a truly independent perspective. It helps stakeholders navigate the complexities of drug innovation, regulatory pathways, and global supply strategies. Recent high-impact analyses on the site are hitting industry pain points head-on—perfect reading for anyone in biopharma.
1. PD-1/PD-L1 Inhibitors: Demystifying the Core Mechanisms of Cancer Immunotherapy
One of the biggest challenges in oncology remains tumor immune evasion. DengYueMed’s latest feature article, “How Do PD-1/PD-L1 Inhibitors Work? Mechanisms of Cancer Immunotherapy Explained” (published March 31, 2026), provides a clear, comprehensive breakdown: Tumor cells express PD-L1, which binds to PD-1 on T cells, suppressing T-cell proliferation and killing activity and leading to T-cell exhaustion. PD-1/PD-L1 monoclonal antibodies (such as Penpulimab and Enlonstobart) block this interaction, reactivating T cells, restoring anti-tumor immunity, and remodeling the tumor microenvironment through increased cytokine release and enhanced immune cell infiltration.
The piece traces the evolution from late-line monotherapy to first-line combinations with chemotherapy, radiotherapy, or targeted therapies, while highlighting practical guidance on managing immune-related adverse events (irAEs). Whether you’re an oncologist, business development professional, or investor exploring next-generation PD-1 combination strategies, this is essential reading. Read the full analysis here: How Do PD-1/PD-L1 Inhibitors Work?
2. NMPA vs FDA Drug Approval: Key Differences Between China and US Regulatory Systems
For global pharma teams, the eternal question is “speed versus rigor.” DengYueMed’s in-depth comparison “NMPA vs FDA Drug Approval: Key Differences Between China and US Regulatory Systems” (March 27, 2026) contrasts the two systems’ philosophies:
- FDA: Prioritizes large-scale randomized controlled trials (RCTs) and long-term safety data, with pathways like Fast Track and Breakthrough Therapy.
- NMPA: Emphasizes speed with controlled flexibility through priority review, conditional approval, and expedited channels for urgent overseas drugs—especially in oncology and rare diseases.
This has led to the unique “China-first” phenomenon where certain innovative therapies reach patients in China ahead of the US. The article also explores real-world implications for cross-border procurement, supply chain planning, and pricing strategies.
Full comparative analysis and case studies available here: NMPA vs FDA Drug Approval
3. Rare Diseases Breakthrough: Global Advances in PNH Complement Inhibitors – From IV to Oral Revolution
Paroxysmal nocturnal hemoglobinuria (PNH) is an ultra-rare disease with significant patient numbers in populous markets like China. DengYueMed’s global overview “New Breakthroughs in Clinical Research on Paroxysmal Nocturnal Hemoglobinuria (PNH) from a Global Perspective” (March 18, 2026) traces the evolution of complement inhibitors:
From the first C5 inhibitor eculizumab (2007) and long-acting ravulizumab, to C3 inhibitor pegcetacoplan, and the highly anticipated oral Factor B inhibitor iptacopan (with positive Phase III APPLY-PNH and APPOINT-PNH data in 2026). Iptacopan has demonstrated significant hemoglobin increases, reduced transfusion dependence, near-normal LDH levels, and—crucially—a shift from intravenous to convenient oral dosing, dramatically improving patient compliance.
The article underscores how PNH has transformed from a life-threatening condition into a manageable chronic disease, offering hope for functional cures across the rare-disease field.
Explore the complete global perspective and latest clinical breakthroughs: PNH Complement Inhibitors Global Breakthrough
Why Choose DengYueMed?
- True Independence: No corporate sponsorship—pure intelligence-driven analysis.
- Practical Focus: Full coverage of FDA/EMA/NMPA pathways plus real-world cross-border supply chain execution.
- Timeliness: Weekly updates on oncology, rare diseases, regulatory changes, and supply chain trends.
Whether you work in pharmaceutical market access, cross-border distribution, investment, or clinical practice, DengYueMed delivers the “one-stop” global pharmaceutical intelligence you need.
Visit DengYueMed today: https://dengyuemed.github.io/ Explore categories: Rare Diseases | Oncology | Regulatory | Supply Chain
In an era of information overload, what matters is distilled, independent insight. DengYueMed is emerging as the indispensable intelligence partner for global pharmaceutical professionals.
-
- DengYue Medicine
- 2 posts
Posted in the topic Recombinant Human Albumin Injection (Oryza Sativa): A Breakthrough for Hypoalbuminemia in Cirrhosis in the forum Off-Topic DiscussionsNovember 21, 2025 12:22 AM PST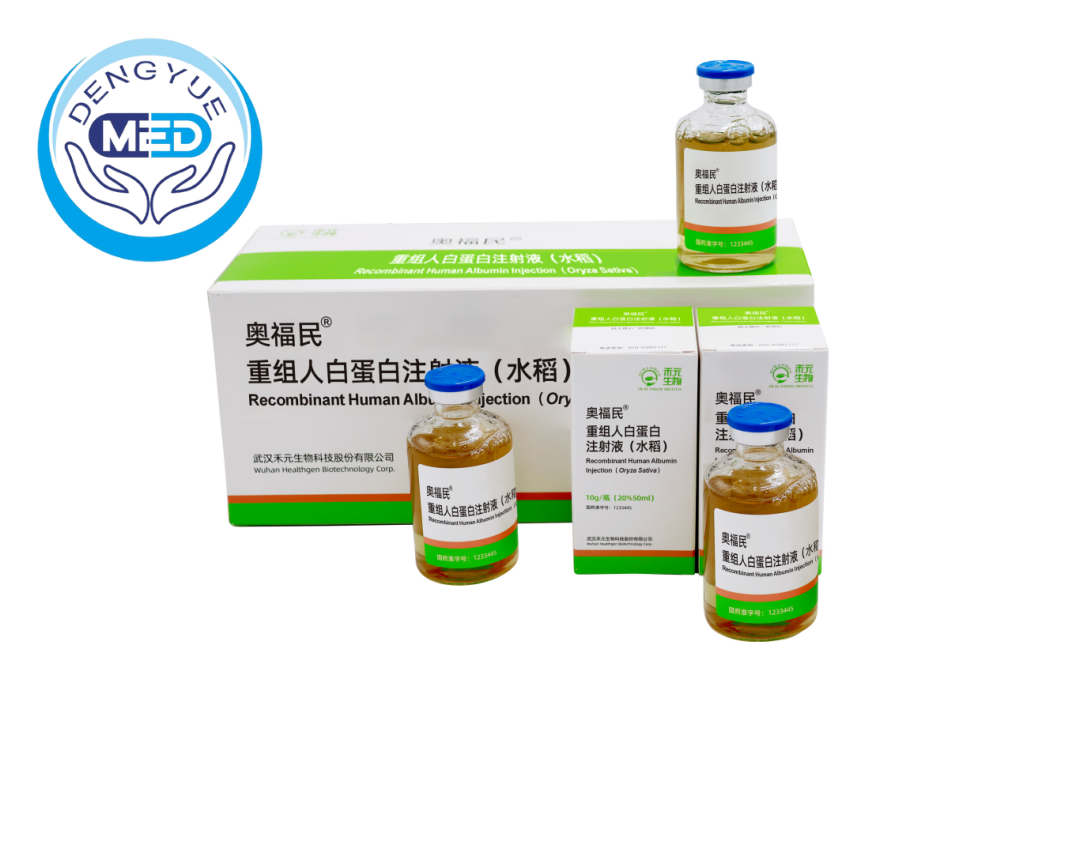
In the field of modern hepatology, cirrhosis continues to pose significant challenges, particularly when accompanied by hypoalbuminemia (serum albumin levels ≤30 g/L), which can lead to complications like ascites and edema, drastically affecting patients' quality of life. Traditional human serum albumin (HSA) derived from plasma has long been the standard treatment, but it faces limitations in supply and risks of blood-borne pathogens. Enter Recombinant Human Albumin Injection sourced from Oryza Sativa (rice)—a plant-based innovation known as OsrHSA—that promises a safer, more scalable alternative for managing hypoalbuminemia in cirrhosis patients.
Understanding Recombinant Human Albumin Injection (Oryza Sativa)
Developed by Wuhan Healthgen Biotechnology Corp., this product leverages genetic engineering to express human serum albumin in rice seeds (Oryza Sativa), using the plant as a natural bioreactor for efficient, large-scale production. Unlike plasma-derived HSA, OsrHSA eliminates dependency on human donors, with production yields reaching up to 10.58% of total soluble protein in rice seeds. This approach not only cuts costs but also removes viral contamination risks. The product received approval from China's National Medical Products Administration (NMPA) in July 2025, with its primary indication being hypoalbuminemia associated with cirrhosis.
The manufacturing process involves inserting the human albumin gene into rice, resulting in a recombinant protein with over 99% purity that is structurally and functionally equivalent to natural HSA. It supports key roles such as maintaining oncotic pressure, drug transport, and fluid balance, making it ideal for clinical use in liver diseases.
Clinical Evidence: Proven Safety and Efficacy
A pivotal multicenter, randomized, double-blind trial (NCT04835480) assessed OsrHSA's performance against plasma-derived HSA in decompensated cirrhosis patients with ascites and hypoalbuminemia (≤30 g/L). Patients received daily intravenous doses of 10g or 20g until serum albumin reached ≥35 g/L, for up to 14 days. OsrHSA demonstrated non-inferiority, with an efficacy rate of 76% in elevating albumin levels, comparable to the control group's 75.6%. Secondary outcomes, including time to target levels, colloid osmotic pressure improvements, and ascites reduction (measured by abdominal girth and weight), showed no significant differences between groups.
Safety profiles were equally favorable: no serious drug-related adverse events, with overall adverse event rates at 66.2% for OsrHSA versus 66.7% for plasma HSA, and minimal immunogenicity. Additional studies, including a Phase Ib trial, further support OsrHSA's role in treating ascites in cirrhosis, highlighting its potential to address global albumin shortages.
Ongoing research, such as NCT06355479, continues to evaluate OsrHSA's efficacy in hypoalbuminemia related to hepatic cirrhosis, reinforcing its therapeutic promise.
The Advantages of Plant-Derived Albumin
For cirrhosis patients prone to recurrent issues like hypovolemia and shock, OsrHSA offers rapid volume expansion and hemodynamic stability without the constraints of plasma sourcing. China's albumin market, the world's largest, consumed over 1,000 tons in recent years, with heavy import reliance. Healthgen's current annual production capacity stands at 10 tons of HSA stock solution, with expansions planned to reach 13 million doses by mid-2026 through new facilities. This scalability could reduce import dependency and make treatment more accessible worldwide.
DengYueMed: Facilitating Global Access to Innovative Therapies
As Chinese innovative drugs gain traction globally, companies like DengYueMed (Hong Kong DengYue Pharmaceutical Co., Limited) play a crucial role in bridging markets. Founded in 2019, this licensed Hong Kong-based wholesaler specializes in the import and export of novel, specialty, and rare drugs, focusing on oncology and chronic diseases like liver conditions. With expertise in cold-chain logistics, professional storage, and global distribution, DengYueMed ensures safe, compliant delivery of cutting-edge therapies. They actively support the export of recombinant proteins and are positioned to help products like OsrHSA reach international patients, contributing to the boom in Chinese pharma exports.
Looking Forward: A New Era in Liver Disease Treatment
Recombinant Human Albumin Injection (Oryza Sativa) marks a pivotal shift in biotechnology, addressing supply bottlenecks and enhancing safety for hypoalbuminemia in cirrhosis. With accumulating clinical data and expanding production, this rice-derived solution is set to become a cornerstone therapy. Patients facing these challenges should consult healthcare providers and explore reliable distributors like DengYueMed for access to such innovations. The future of liver care is rooted in sustainable science!